BIOMÉRIEUX CLARION™
Stewardship from Lab to Leadership
Clinical Informatics for Lab Insights

Clinical Informatics for Lab Insights

Clinical Informatics for Stewardship Programs

Clinical Informatics for Visualizing COVID-19

Stewardship from Lab to Leadership

Stewardship from Lab to Leadership

Optimize Efficiency and Outcomes with Data-driven Infectious Disease Management
CLARION is a secure, cloud-based clinical informatics software-as-a-service that connects and integrates disparate data into a single management application. CLARION gives you a holistic view of relevant lab and clinical data across departments, processes, and vendors and translates this data into actionable insights visualized on easy-to-use dashboards.
Utilization of our intuitive CLARION platform provides the capability to track and improve metrics that can impact patient care and outcomes. We leverage our expertise in microbiology and infectious diseases to help health systems understand their current processes then visualize and analyze their data.
CLARION Addresses Key Issues Healthcare Organizations Face in Infectious Disease Management:
CLARION Complete | Analysis and Deeper Insights
Advanced insights into lab workflow and diagnostic stewardship.
-
 Save time
Save time
-
 Focus on quality
Focus on quality
-
 Provide meaningful insights to clinicians
Provide meaningful insights to clinicians
-
 Anticipate future trends and resistance patterns
Anticipate future trends and resistance patterns
Dashboards Include:
Infectious Disease Management
- Advanced Antibiograms in Clinical and Laboratory Standards Institute (CLSI)
- Susceptibility Analysis
- MDRO Reports
- MIC Distribution & Surveillance
- Pathogen Specific Dashboards (Influenza, A&B, RSV, Group A Strep)
- Testing Analysis for STDs, C. difficile & TB
- Alerts
Micro Lab Workflow
- Complex Blood Culture Data Analysis
- VITEK® MS Spot Analysis
- Urine Contamination
- VIDAS® Assay Analysis
- Alerts
CLARION Stewardship | Diagnostic Stewardship
Clinical insights focused on the key elements of a stewardship program from consolidated data and analytics. A comprehensive module that provides a holistic view of infectious disease management and diagnostic stewardship by integrating feeds from multiple data sources. Includes all of the dashboards in CLARION Complete, allowing you to derive clinical insights from consolidated data and analytics.
-
 Gain deep understanding of diagnostic deployment
Gain deep understanding of diagnostic deployment
-
 Impact clinical outcomes and empower physician confidence
Impact clinical outcomes and empower physician confidence
-
 Tailor diagnostic and treatment decisions to your emerging resistance patterns
Tailor diagnostic and treatment decisions to your emerging resistance patterns
-
 Focus on what is important to your stewardship program
Focus on what is important to your stewardship program
Dashboards Include:
Infectious Disease Management
- Antimicrobial Stewardship Analysis
- Diagnostic Stewardship Analysis
- Syndromic Antibiogram
- Combination Antibiogram
- Targeted Phenotype Antibiogram
- Outcomes Benchmarking
- Other Pathogens Dashboards (Lyme, Measles, TB)
- Geomapping
Micro Lab Workflow
- Lab Turn-Around-Time
- Test Volume Analysis
- BIOFIRE® FILMARRAY® Assay Analysis
- Geomapping
CLARION Value: VIDAS® B·R·A·H·M·S PCT™ | Measure Impact on Outcomes
A deep look at the utilization of VIDAS® B·R·A·H·M·S PCT™, tied to antibiotic usage and patient outcomes that integrates additional data from the laboratory, Pharmacy, Admit, Discharge, Transfer (ADT) system, and Electronic Health Records (EHR). This module aims to optimize biomarker utilization, view clinician prescribing, and response.
-
 Benchmark and monitor best practices
Benchmark and monitor best practices
-
 Facilitate informed clinical discussions and targeted education
Facilitate informed clinical discussions and targeted education
-
 Quantify how diagnostic stewardship drives value-based healthcare
Quantify how diagnostic stewardship drives value-based healthcare
Dashboards Include:
- PCT Usage by Indication
- Clinician-specific Utilization and Outcomes Measures
- Outcomes Analyses (e.g., Length of Stay)
- Pharmaceutical Utilization Dashboards: Days of Therapy, Length of Therapy Analysis
- Mortality Analysis
- Clinician-specific Results Response
Unprecedented Demands Require Unprecedented Solutions
Keeping vigilant, and defeating the COVID-19 outbreak, is a challenge to everyone in healthcare. The unprecedented speed of the outbreak, and already heavily burdened IT departments, make it difficult to quickly develop and deploy innovative COVID-19 solutions in our nations' hospitals.
History Fighting Infections
bioMérieux Inc., known for its CLARION stewardship solutions, has released a new COVID-19 Surveillance Dashboard on a no-obligation, 30-day trial.* Hosted in the cloud, without the added demands on the IT department, our solution can be ready in less than 48 hours.
Right now, speed and agility are essential. Leveraging existing bioMérieux cloud technology, your lab data will be processed by the CLARION team and incorporated into a secure, individualized, and password-protected solution accessible via Web and mobile devices.
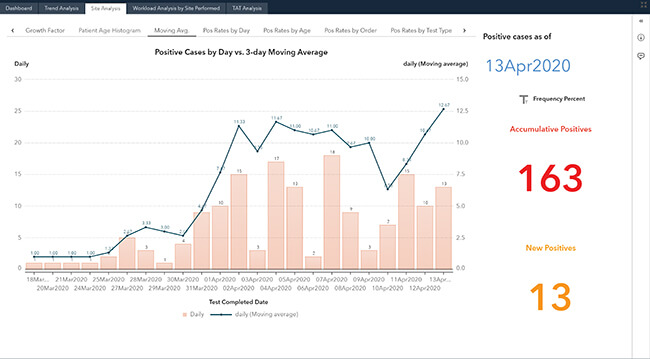
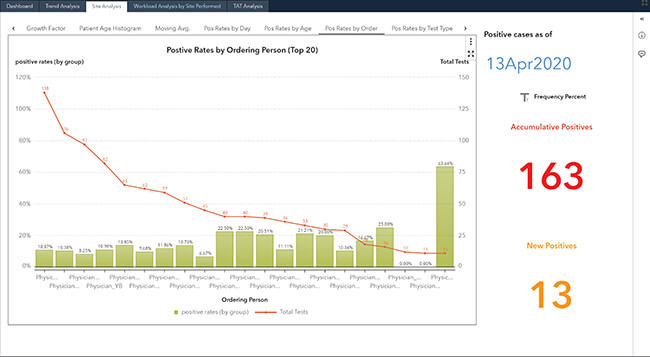
Visualize COVID-19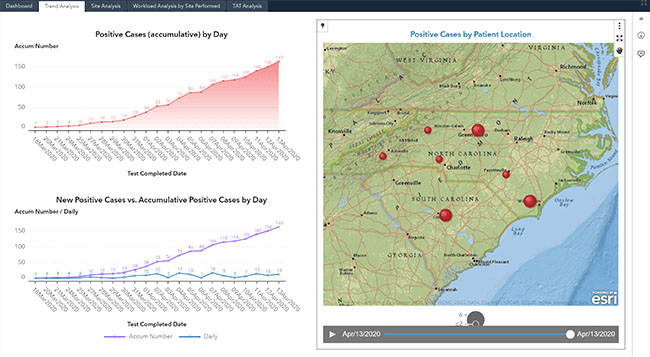
Hospitals not only treat their inpatients, but have a requirement to monitor and serve their ambulatory communities. To meet those requirements, our solution delivers a highly visual set of surveillance dashboards from your data using pre-defined data elements, with an ability to evolve to subscribers' needs. The dashboards provide analysis and geolocation to track cases by county, city, and zip code for meeting the unprecedented demand during the pandemic. These dynamic COVID-19 Surveillance Dashboards are designed to be intuitive and iterative.
Schedule a Consult
If you have additional questions, or would like to schedule a consultation, contact us at Clarion@bioMerieux.com. Our CLARION Team is comprised of world-class data integration experts, visualization experts, a clinical data scientist, and healthcare professionals who are ready to help put our solutions to work for you.
*The initial trial period is 30 days, after which the price is $500/month with a minimum 12 month commitment. Signed SaaS (software-as-a-service) agreement is required at start of the 30 day trial.

PRN 055974 Rev 01.A
bioMérieux takes the security of your data and patient health information extremely seriously. For this reason we have carefully chosen technology partners that have a long track record of data security and are approved for use with patient data.
Our Integration Engine is an FDA Certified non-device MDDS (under the 21st Century Cures Act) and compliant to FDA 21 CFR Part 820.
CLARION is hosted on a secure private cloud that is compliant to HIPAA and GxP (FDA 21 CFR Part11). As a secure, cloud-based service, CLARION is designed to comply with the HIPAA Privacy Rules, Safe Harbor method for de-identification of data:
- De-Identify Data – takes place in the Integration Engine inside your firewall.
- Securely Transmit Data – with different encryption methods and a complete audit trail of all data transfers, you have options on how we securely transfer files to the CLARION environment. No PHI is transferred outside your firewall
- Offer Secure Private Cloud Hosting – our secure cloud is compliant to HIPAA and GxP (FDA 21 CFR Part11).
- Deliver Secure User And Administrator Access – authentication and identification also integrates with local administrator’s identity management or Active Directory systems
You can only see your own data, which is stored on a private cloud.
Interested in CLARION?
Fill out the form below to be contacted by a bioMérieux representative, who will provide you with more information on how we can support your evidence-based decision making with CLARION!





